Material below summarizes the article 3D Electron Microscopy Study of Synaptic Organization of the Normal Human Transentorhinal Cortex and Its Possible Alterations in Alzheimer’s Disease, published on June 19, 2019, in eNeuro and authored by M. Domínguez-Álvaro, M. Montero-Crespo, L. Blazquez-Llorca, J. DeFelipe, and L. Alonso-Nanclares.
Highlights
- The majority of synapses are excitatory on spine heads (55%) and have a small disk shape. Nevertheless, a relatively large proportion of excitatory synapses are established on dendritic shafts (37%).
- Excitatory and inhibitory synapses show different preferences for their postsynaptic targets (spines or dendritic shafts).
- In Alzheimer’s disease (AD), morphological synaptic changes and a reduction in the number of synapses targeting spine heads occur.
 |
|
|
Study Question
What is the synaptic organization of the transentorhinal cortex and what are the possible synaptic alterations that occur in Alzheimer’s disease?
How This Research Advances What We Know
Determination of postsynaptic targets, as well as the shape and size of the synaptic junctions, provides critical data about synaptic functionality and circuit organization.
As far as we know, 3D synaptic morphology has never been performed together with the identification of postsynaptic targets in normal human brain samples or in the brain of patients with Alzheimer’s disease (AD). Thus, this study represents the first attempt to unveil the synaptic organization of the neuropil of the human brain at the ultrastructural level.
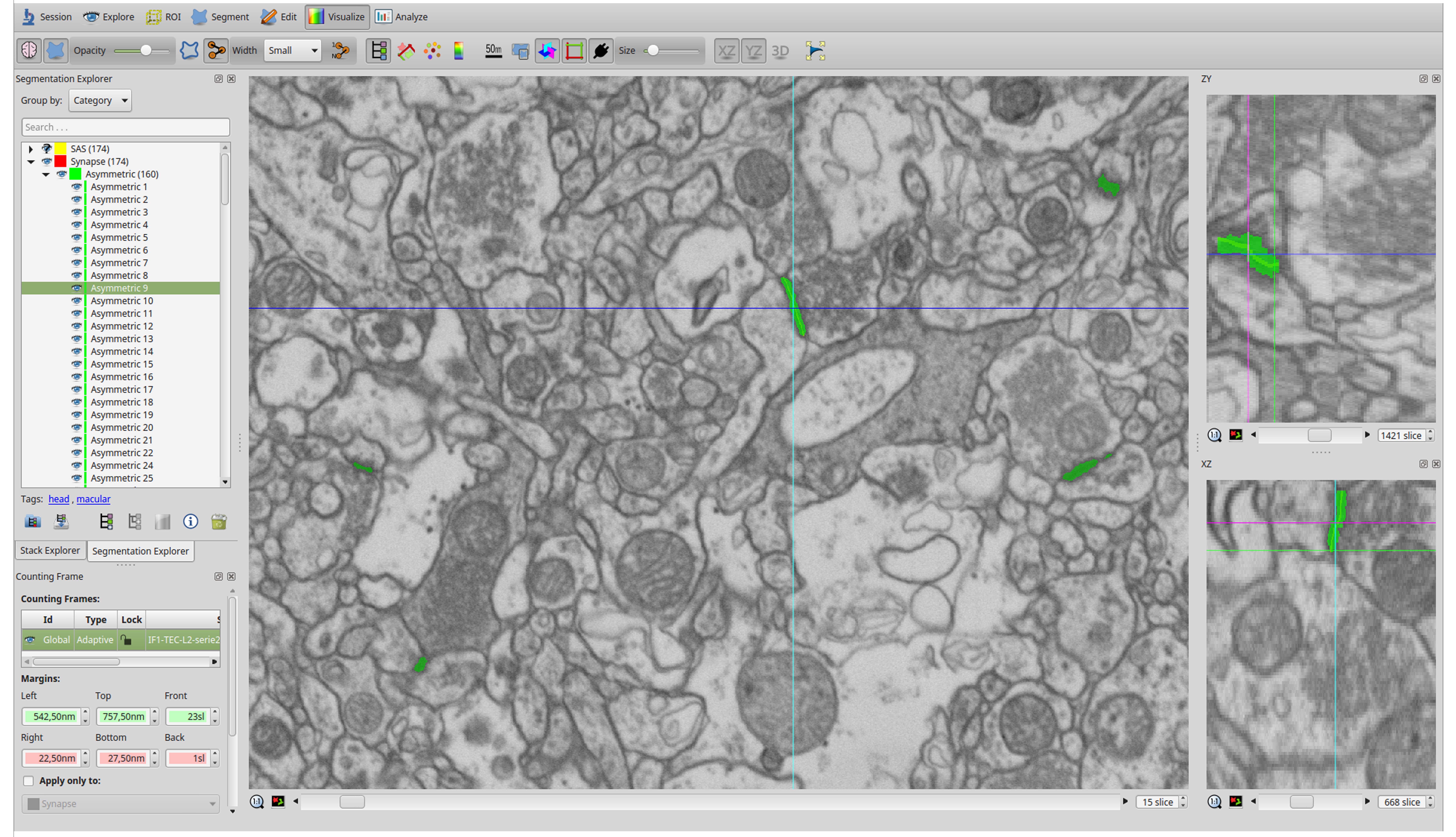
Thanks to 3D electron microscopy (FIB/SEM technology), it is possible to obtain large numbers of 3D reconstructed synapses. This study provides a large new quantitative ultrastructure dataset of the transentorhinal cortex, including both normal and AD cases.
In addition, the morphological synaptic alterations and changes in the postsynaptic targets found in AD may further understanding of the relationship between alterations of the synaptic circuits and the cognitive deterioration observed in these patients.
Experimental Design or Methodology
Human brain tissue samples from five control cases (with no recorded neurological or psychiatric alterations) and five AD patients (time between death and tissue processing was lower than 4h in all samples) were analyzed using FIB/SEM technology. Using this 3D electron microscopy method, we determined the morphology and postsynaptic target (spines or dendritic shafts) of the 4722 identified synapses in the neuropil of layer II of the human transentorhinal cortex. Moreover, we differentiated between excitatory and inhibitory synapses (based on their differential morphology). For data analysis, we applied appropriated statistical analysis and contingency tables.
Results
First, both in control cases and AD patients, excitatory and inhibitory synapses show different preferences for postsynaptic targets: Excitatory synapses have a significant preference for spine heads, while inhibitory synapses have a significant preference for dendritic shafts.
Second, in relation to the shape of synapses, in control and AD patients, the vast majority of synapses have a disk (macular) shape, followed by synapses with one or more holes (perforated), synapses with indentations in the perimeter (horseshoe-shaped), and synapses with two or more portions (fragmented).
Third, AD patients show an increase in the number of fragmented synapses and a reduction in the number of synapses targeting spine heads.
Interpretation
The large quantitative synaptic ultrastructure dataset of the normal transentorhinal cortex would be useful for comparing with other brain regions when the data become available. The aim would be to try to determine if the same synaptic organization exists in cortical regions that are cytoarchitectonically and functionally different.
Furthermore, this study provides evidence of a reduction in the number of synapses targeting spine heads in AD patients. The loss of spines and, therefore, the loss of synapses and circuits could be related to cognitive decline observed in AD.
Also, we observe morphological synaptic alterations in these patients that could be due to the activation of compensatory mechanisms in response to the synaptic loss that occurs in this cortical region during the progression of the disease. However, it could be possible that this kind of mechanism fails and synaptic activity would fall off.
Visit eNeuro to read the original article and explore other content. Read other summaries of eNeuro and JNeurosci papers in the Neuronline collection SfN Journals: Research Article Summaries.
3D Electron Microscopy Study of Synaptic Organization of the Normal Human Transentorhinal Cortex and Its Possible Alterations in Alzheimer’s Disease. M. Domínguez-Álvaro, M. Montero-Crespo, L. Blazquez-Llorca, J. DeFelipe, and L. Alonso-Nanclares. eNeuro June 2019, 6 (4) 0140–19.2019; DOI: 10.1523/ENEURO.0140-19.2019









