Material below summarizes the article Functional Integration of Adult-Born Hippocampal Neurons after Traumatic Brain Injury, published on September 14, 2015, in eNeuro and authored by Laura E. Villasana, Kristine N. Kim, Gary L. Westbrook, and Eric Schnell.
Although the majority of neurons in mammalian brains are generated during embryologic development, the production of new neurons continues throughout life in at least two brain regions in a process known as adult neurogenesis. These adult-born neurons integrate into neuronal circuits and are believed to play an important role in brain function. In the hippocampus, new granule cells are born in adult animals and contribute to learning and memory. Interestingly, the production of these adult-born granule cells increases markedly after certain forms of neuronal injury, including seizures, strokes, and traumatic brain injury (TBI). It has been hypothesized that these new neurons might play a role in recovery.
Most of the studies that have analyzed post-injury neurogenesis involved identification of neurons using immunohistochemical markers, but few studies have characterized the functional properties of these cells. Prior physiologic studies have shown that neurons born after seizures are aberrantly integrated into the hippocampal network, so we wondered whether the neurons born after TBI would have functional alterations as well. The functional state of these neurons could have significant implications for their contribution to information processing after injury.
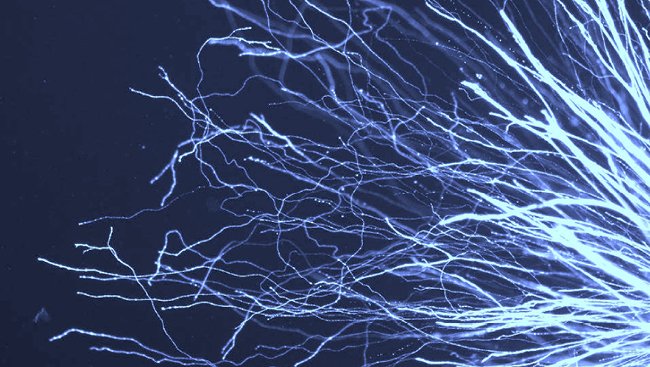
To address this issue, we used a controlled cortical impact model of TBI in transgenic mice, in which fluorescent proteins could be selectively expressed in adult-born neurons. This allowed us to characterize the morphologic development of the new neurons born after TBI, and to assess their functional integration into neuronal circuits through the use of whole-cell recording techniques.
Cells born after TBI migrated abnormally across the granule cell body layer in the hippocampus, and had abnormally branched dendrites when compared with age-matched adult-born granule cells in healthy brains. These changes became apparent during the early maturation of these granule cells, and persisted as the cells matured. However, despite their grossly abnormal appearance, we found that these cells did not notably accelerate or otherwise alter their synaptic integration into the brain. Cells born after TBI established normal overall levels of both excitatory and inhibitory synaptic inputs, and their inputs from cortical afferents possessed both normal pre-synaptic functional properties and a balance of post-synaptic receptor expression. Thus, these cells were both functional and integrated into the hippocampal network.
What are the implications of these apparently disparate results? The "good news" is that cells born after TBI become integrated into circuits and survive to maturity, thus likely taking part in hippocampal information processing, consistent with a role in post-injury cognitive recovery. However, the morphologic aberrations that we found in these cells could still be "bad news," as the altered dendritic branching has potential implications for the relative weighting of inputs representing different information streams. Since the cells born in injured brains have more dendrites in proximal regions of their dendritic trees, they might be predominantly influenced by the intrinsic inputs that synapse in that region of the hippocampus, and might be correspondingly less responsive to cortical inputs that synapse more distally. We did not test the relative input strengths between different information streams, so the possibility that the differences we noted in the morphology of these cells have an adverse functional implication remains. We hope our future work will be able to address this possibility.
Visit eNeuro to read the original article and explore other content. Read other summaries of JNeurosci and eNeuro papers in the Neuronline collection SfN Journals: Research Article Summaries.
Functional Integration of Adult-Born Hippocampal Neurons after Traumatic Brain Injury. Laura E. Villasana, Kristine N. Kim, Gary L. Westbrook, Eric Schnell. eNeuro Sep 2015, 2 (5) DOI: 10.1523/ENEURO.0056-15.2015









