Material below is adapted from the SfN Short Course Distinct Molecular Programs Define Human Radial Glia Subtypes During Human Cortical Development, by Tomasz J. Nowakowski, PhD, and Alex A. Pollen, PhD. Short Courses are day-long scientific trainings on emerging neuroscience topics and research techniques held the day before SfN’s annual meeting.
Multicellular organisms were able to evolve in part because of the capability of cells to take specialized shapes and perform varied functions. Understanding how cells diversify their characteristics has become simpler with the rise of single-cell RNA sequencing, which allows scientists to learn more about the genes and pathways active in an individual cell. Researchers can apply this technique to learn more about development of the human nervous system.
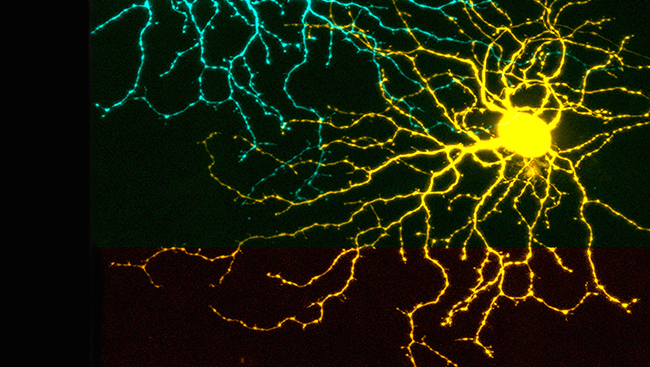
During human development, the many types of cells in the brain all arise from what begins as a homogeneous tissue layer, called a neuroepithelium. Through a series of cellular divisions, induction events, and movements, eventually the brain emerges, containing diverse cell types. Two neuronal stem cell types are the ventricular radial glia (vRG), which are located in the ventricular zone, and the outer radial glia (oRG), which reside in the outer subventricular zone of the developing cortex.
In an effort to identify differences between vRG and oRG, one research team sequenced RNA from single cells that were captured from the ventricular zone and outer subventricular zone of the developing human cortex. They analyzed the transcriptomes of the cells they sequenced and compared them to known transcriptomes of radial glia, to determine which cells were likely to be radial glia.
Then, the researchers more closely examined the gene expression profiles of 107 potential radial glia. They found variation in genes involved in DNA replication, cell-cycle regulation, and mitosis, as well as in position of the cells within the cortex. Based on these distinctions, they identified two subgroups of cells, which were predicted to correspond to vRG and oRG.
Next, the research team defined a group of candidate marker genes, based on genes that were likely to be specific to one, the other, or both radial glia subtypes. They investigated their candidate gene markers with in situ hybridization and immunostaining, and found that predicted vRG markers were most strongly expressed in the ventricular zone, and that predicted oRG markers were most strongly expressed in the outer subventricular zone. They also used time-lapse imaging to observe how cell movements related to their predicted gene markers.
The researchers identified a relationship between cell shape, behavior, location, and the oRG and vRG molecular markers. Many of the confirmed markers were functionally relevant, in that they were genes involved in pathways that the two types of radial glia activate to perform their proliferative functions or that they use to cope with the challenges in their specific cortical niche. The analysis also revealed that oRG cells in the human cortex are capable of generating vast numbers of progeny—a finding that was surprising based on previous studies in mice, whose radial glia generate far fewer daughter cells.
Exploring the gene expression profiles of individual cells captured from tissue containing many cell types is now possible and presents a powerful strategy for understanding groups of cells. Plus, relating gene expression to morphology, behavior, and location within a tissue provides insight into how cells function and contribute to the development of complex systems like the brain. An exciting future direction for this work will be to couple single-cell RNA sequencing with high-throughput analyses of cellular location, behavior, and shape, as well as of neuronal activity.







