QR2 Modulates Insular Cortex Redox to Enable Novel Taste Memory Formation
Material below summarizes the article Muscarinic-Dependent miR-182 and QR2 Expression Regulation in the Anterior Insula Enables Novel Taste Learning, published on March 26, 2020, in eNeuro and authored by Nathaniel L. Gould, Alina Elkobi, Efrat Edry, Jonathan Daume, and Kobi Rosenblum.
Highlights
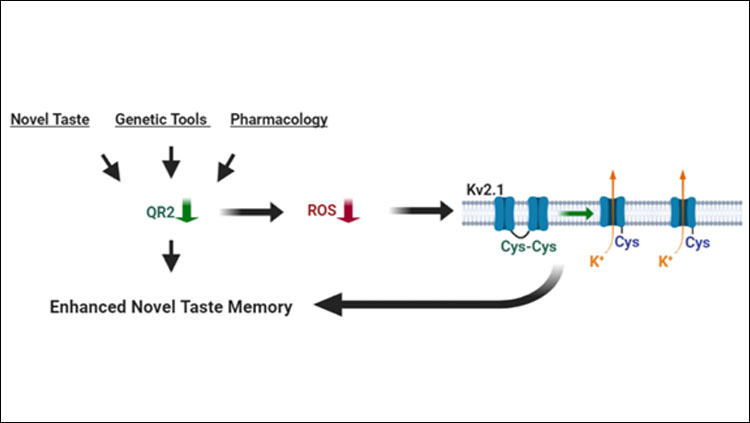
- QR2 is involved across species in the important process of forming new memories of unfamiliar tastes, enabling animals to learn about safe or harmful foodstuffs.
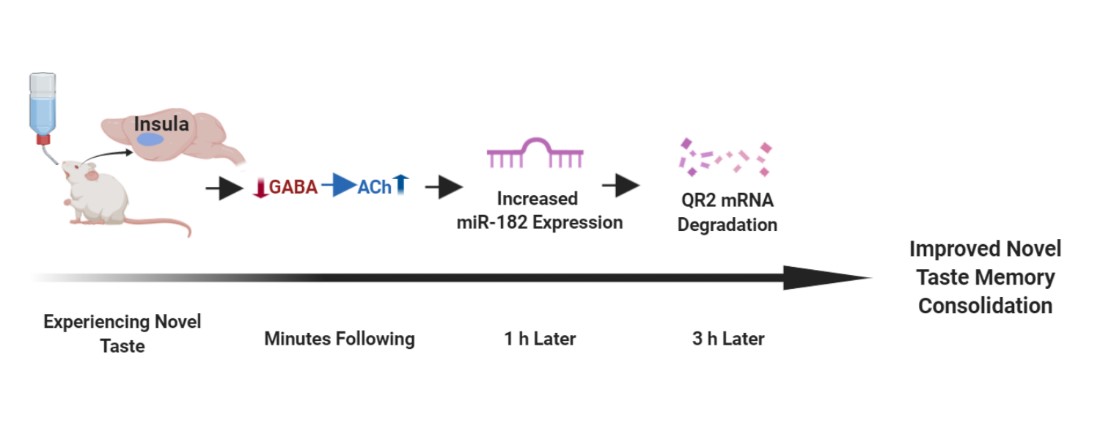
- It does so by being removed from the anterior insular cortex (aIC), via the destabilization of its mRNA by miR182, which is itself dependent on local disinhibition of acetylcholine (ACh) release.
- Removal of QR2 in the aIC reduces reactive oxygen species (ROS) there, affecting the potassium channel Kv2.1, which results in a stable long-term memory of a newly experienced taste, a process that deteriorates with age and disease.
 |
|
|
Study Question
Animals, including humans, form a long-term memory of new incidental information in order to optimize future behavior. We previously found QR2 in the insular cortex is a removal constraint on new taste memory formation. We therefore asked whether this happens across species, how it happens, and what it causes to affect memory.
How This Research Advances What We Know
Usually, we think the stronger our memory, the better. However, biology distinguishes between important and trivial information by filtering it at different stages of learning.
Very little is known about removable memory constraints in general during this process, and about QR2 in the brain in particular. This little-understood enzyme was surprisingly found to be involved in the formation of important, novel taste memories. It has been shown that QR2 is removed from the primary taste cortex, the anterior insula, by the neurotransmitter acetylcholine in order to allow a robust new taste memory.
Here, we set out to see how exactly a reductase, ostensibly meant to be clearing away harmful quinones, is linked to learning and memory. We therefore determined whether QR2 is, in fact, a functional reductase, how it is controlled by acetylcholine in the cortex — and to what outcome, in order to establish its functional attributes in the brain.
Experimental Design or Methodology
Rats or mice were randomly allocated groups, following acclimatization to the animal facilities and handling. Animals were kept in enriched cages, and some had cannulas implanted to the aIC, in order to enable local delivery of drugs. All taste experiments were done by first teaching animals to drink from pipettes, to enable measurement of liquid consumption. Then, they were given novel tastes to drink. After 24 or 48 hours, their memory for the taste was tested, by giving them a choice between water and the tastant (saccharin or NaCl). Later, we analyzed levels of different brain RNAs and proteins.
All cell-based experiments were carried out in human embryonic kidney (Hek) cells. ROS and enzyme activity assays based on synthetic co-factor oxidation were carried out using a fluorimeter. HPLC was used to measure the absorbance of QR2 product formation.
Results
Is it a cross-species phenomenon? QR2 suppression in the aIC following novel taste consumption occurs in both rats and mice.
Can we enhance memory artificially? Removal of QR2 prior to- or inhibition of QR2 after learning enables improved taste memory.
What is upstream of QR2 removal? Inhibiting QR2 reversed muscarinic ACh receptor (mAChR) antagonism mediated amnesia, making QR2 an important target of ACh in taste memory formation. Furthermore, GABAA receptor (GABAAR) antagonism mimics ACh release in that it suppresses QR2 expression and enhances taste memory, but prior mAChR antagonism prevents these effects. Therefore, local aIC GABAAR inactivation allows ACh efflux, leading to mAChR activation and QR2 suppression. Additionally, QR2 is not suppressed via reduced transcription from the genome. Rather, QR2 is targeted for suppression by miR182, which is inversely upregulated by the same stepwise GABAAR inactivation and mAChR activation that follows novelty.
Does QR2 affect ROS in the brain? Fluorescent synthetic cofactors used to measure total quinone reductase activity in the brain showed sensitivity to QR2 inhibition, and small molecules in the brain are able to act as cofactors for purified QR2. Cellular QR2 and NQO1 activity outcomes showed ROS are increased by NQO1 inhibition but decrease upon QR2 inhibition.
What is downstream from QR2 removal that explains better memory? In the brain, QR2 suppression or inhibition results in reduced oxidation of voltage gated potassium channel Kv2.1, indicating a reduction in ROS which presents a plausible scenario that QR2 affects intrinsic neuronal properties and thus function.
Interpretation
These results demonstrate for the first time a function for QR2 in the brain, and describe upstream and downstream mechanisms and targets. By acting as a reductase in the aIC, QR2 generates physiological levels of ROS, which upon its suppression are removed. This is a form of oxidative eustress, which via redox modulation in the cortex helps novel memory formation.
This unusual involvement of a reductase in novel memory formation ties in with the known effects of ACh depletion (and QR2 overexpression) that occur in Alzheimer’s disease — inflammation and anterograde amnesia. It is therefore possible that QR2 may present a new way towards tackling such pathologies. However, it is unknown how QR2 redox modulation affects neurons, whether it is a general mechanism or just found in the aIC following taste learning. The full spectrum of QR2 sensitive targets, beyond Kv2.1, also require identification. This exciting new avenue merits further investigation.
Visit eNeuro to read the original article and explore other content. Read other summaries of eNeuro and JNeurosci papers in the Neuronline collection SfN Journals: Research Article Summaries.
Muscarinic-Dependent miR-182 and QR2 Expression Regulation in the Anterior Insula Enables Novel Taste Learning. Nathaniel L. Gould, Alina Elkobi, Efrat Edry, Jonathan Daume, and Kobi Rosenblum. eNeuro 26 March 2020, 0067-20.2020; DOI: 10.1523/ENEURO.0067-20.2020








