Mouse Models of Fragile X Syndrome Exhibit Subtle Deficits in Auditory Spatial Hearing
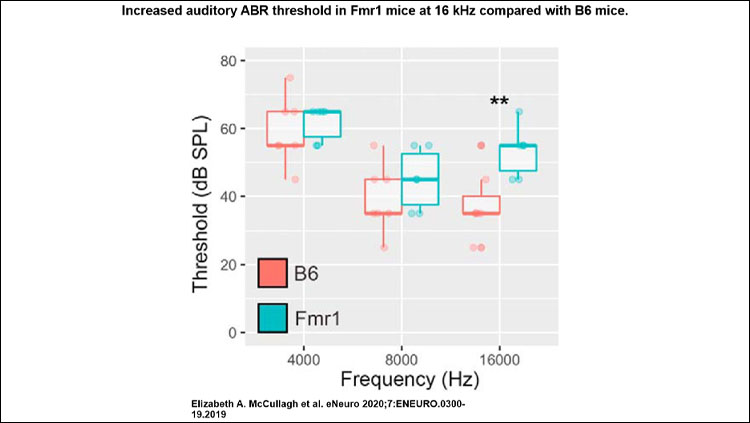
Material below summarizes the article Characterization of Auditory and Binaural Spatial Hearing in a Fragile X Syndrome Mouse Model, published on January 17, 2020, in eNeuro and authored by Elizabeth A. McCullagh, Shani Poleg, Nathaniel T. Greene, Molly M. Huntsman, Daniel J. Tollin, and Achim Klug.
Highlights
- Fragile X Syndrome (FXS) mice have decreased inhibition of acoustic startle responses under several auditory conditions including gaps in noise, large changes in sound source positions, and discrimination of target sounds from background noise compared to wildtype control mice.
- FXS mice took longer to respond to startling sounds in most conditions suggesting additional impairments to pathways underlying when to respond.
- FXS mouse models exhibit similar hearing impairments to humans with FXS.
 |
|
|
Study Question
Do mouse models of FXS, the most common genetic form of autism, exhibit decreased spatial auditory acuity consistent with anatomical and physiological measures of altered auditory brainstem (sound localization) pathways?
How This Research Advances What We Know
While there have been studies demonstrating physiological and anatomical alterations to the auditory brainstem, the area of the brain that first processes sound location, in FXS, no studies have shown whether these alterations actually cause functional behavioral changes to auditory spatial acuity. Disrupted sound location processing could underly auditory hypersensitivity phenotypes seen in patients with FXS.
Experimental Design or Methodology
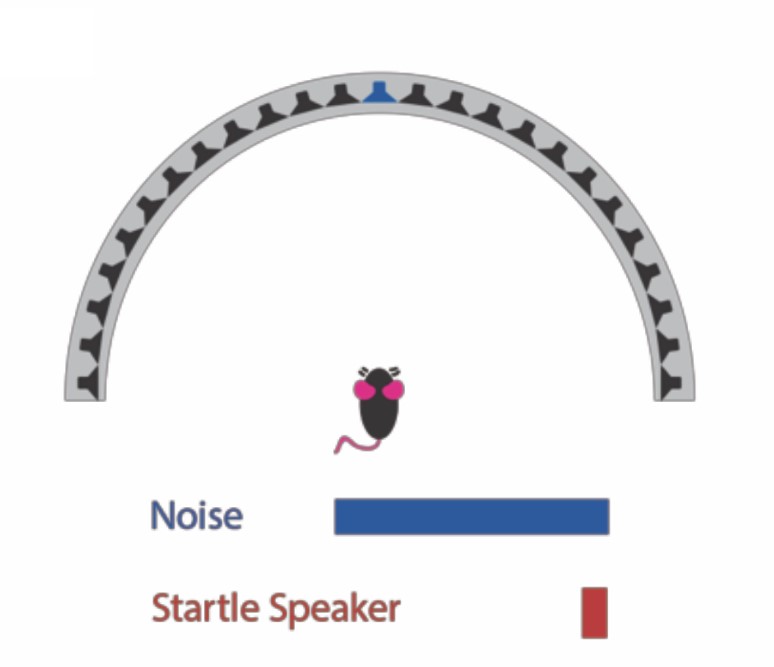
Mice with an FXS mutation were tested under several auditory spatial acuity conditions including temporal gap in noise detection, changes in sound source location, and detecting a signal within background noise. These behavioral tests leverage an animal’s reflexive inhibition of their startle response when given a prepulse cue (the acoustic conditions above) prior to a startling sound. This inhibition of their startle response can then be used to determine the degree to which an animal can detect or respond to the prepulse cue (e.g., a change in sound source location).
In addition to how much they responded to the prepulse, we also measured response latency to the prepulse cue. These tests were performed on both male and female mice, in FXS mutant mice and wildtype controls.
Results
We found that for several of the different prepulse cues the FXS mice took longer to respond to the startle stimulus. In addition, FXS mice showed less inhibition of their startle response compared to wildtypes in gaps in noise, large speaker swap angles, and large angles and loud sounds during the signal in noise task, which indicates less sensitivity to these acoustic stimuli. Overall FXS mice had subtle deficits in auditory spatial hearing compared to wildtypes, with a robust increased latency to respond during many of the PPI conditions.
Interpretation
FXS mice do not have severe spatial hearing deficits but do have impairments in the timing of responses. Despite a rich literature suggesting anatomical and physiological changes to the pathways encoding sound information, our study would suggest that at the behavioral level, the spatial acuity may be largely unaffected. However, these data in FXS mice are largely consistent with what has been seen in FXS patients, suggesting that in terms of the sound localization circuit, the FXS mouse model is similar to humans.
Visit eNeuro to read the original article and explore other content. Read other summaries of eNeuro and JNeurosci papers in the Neuronline collection SfN Journals: Research Article Summaries.
Characterization of Auditory and Binaural Spatial Hearing in a Fragile X Syndrome Mouse Model. Elizabeth A. McCullagh, Shani Poleg, Nathaniel T. Greene, Molly M. Huntsman, Daniel J. Tollin, and Achim Klug. eNeuro 17 January 2020, 7 (1) 0300-19.2019; DOI: 10.1523/ENEURO.0300-19.2019








