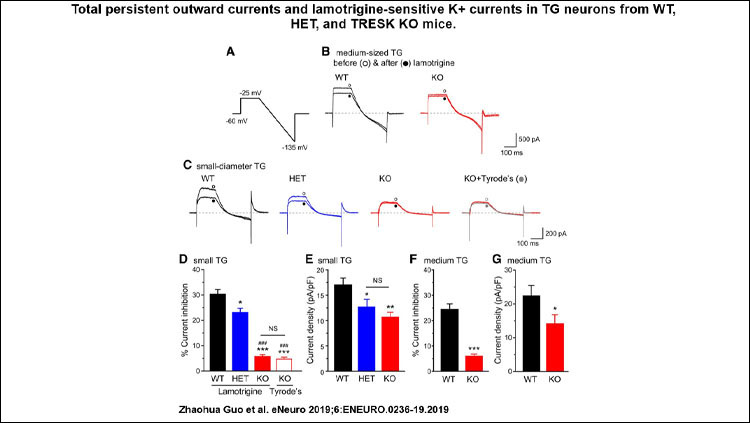
Material below summarizes the article TRESK K+ Channel Activity Regulates Trigeminal Nociception and Headache, published on July 15, 2019, in eNeuro and authored by Zhaohua Guo, Chang-Shen Qiu, Xinghua Jiang, Jintao Zhang, Fengxian Li, Qin Liu, Ajay Dhaka, and Yu-Qing Cao.
Highlights
- We found that loss of TWIK-related spinal cord K+(TRESK) in all trigeminal ganglia (TG) neurons preferentially increased the intrinsic excitability of small-diameter TG nociceptors that express neuropeptide CGRP (calcitonin gene-related peptide) or TRPM8 channels.
- Compared with wild-type (WT) controls, TRESK knockout (KO) mice exhibited more robust trigeminal pain, especially headache-like behaviors, but displayed normal body and visceral pain responses, indicating that genetic loss of TRESK significantly increases the chance of developing headache.
- Our study highlighted some exquisite differences between the head pain-sensing TG neurons and body pain-sensing dorsal root ganglion (DRG) neurons in response to ion channel defects, supporting a causal relationship between defective TRESK channel and higher migraine susceptibility.
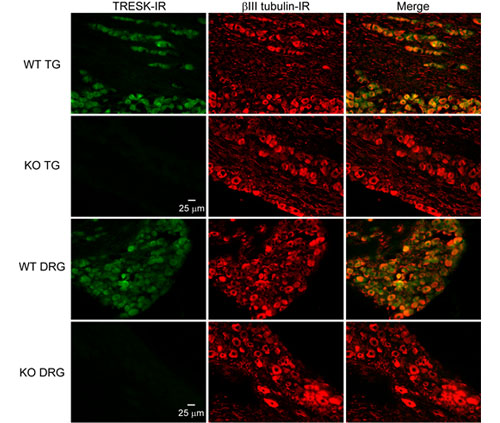
Representative images of TG and DRG sections from adult WT and TRESK KO mice double stained with antibodies against TRESK and βIII tubulin. βIII tubulin-IR is present in all neurons. TRESK-IR is present in almost all neurons in the WT sections. There is no TRESK-IR in the KO sections. Adapted from Figure 1, Guo et al., eNeuro, 2019.
Study Question
In this study, we used TRESK global knockout (KO) mice to investigate how endogenous TRESK activity regulates trigeminal nociception. Does endogenous TRESK activity regulate the intrinsic excitability of all or subpopulation(s) of TG neurons? Does genetic TRESK dysfunction selectively enhance trigeminal pain at the behavioral level?
How This Research Advances What We Know
Migraine headache is one of the most common neurovascular disorders and the second leading cause for disability worldwide, affecting 12% of the general population. Recurring headache, which lasts between 4 and 72 hours, is highly debilitating, poorly understood, and difficult to treat. Elucidating the mechanisms underlying migraine headache is important for the development of better and safer treatments.
Mutations that generate defective TRESK channels have been found in migraine patients, but the causal relationship between TRESK defect and migraine headache has not been established. TRESK channels are expressed in TG neurons sensing head pain as well as in DRG neurons sensing body pain. Why are TRESK mutations associated with migraine and not with any types of body pain?
Experimental Design or Methodology
We analyzed the TRESK KO mice, which do not express TRESK channel in any cells from neonatal to adult, by immunostaining TG and DRG sections from WT and KO mice with an antibody that recognizes mouse TRESK protein.
Using patch-clamp recording, we compared TRESK-like current in TG and DRG neurons from WT and KO mice, as well as intrinsic excitability.
To address if genetic loss of TRESK affected the excitability of dural afferent neurons, the primary sensory neurons in the trigeminovascular pathway subserving headache. Fluoro-Gold (FG) was used to retrogradely label dural afferent neurons in adult mice. We compared the size distribution and intrinsic excitability of FG labeled neurons of WT and KO mice.
In addition, headache-related behaviors were observed in both WT and KO mice that recovered from craniectomy by dural application of either saline or IScap (a mixture of inflammatory mediators).
Results
None of the TG or DRG neurons from KO mice had TRESK-like current. Interestingly, loss of TRESK-like current led to very different consequences in TG and DRG neurons.
In TG neurons, absence of TRESK channel made one-third hyper-excitable, i.e. they could be activated by a smaller depolarizing current, and when activated, they generated more action potential. Some TG neurons from KO mice even spontaneously generated action potentials. Notably, many of these neurons expressed neuropeptide CGRP. Because CGRP is released during migraine headache, and blocking CGRP can present migraine in many patients, we hypothesized that KO mice may be more likely to develop headache-like behaviors due to the hyper-excitation of CGRP-expressing TG neurons.
The generation of headache usually starts with the activation of TG neurons that project their axons to meninges. To mimic this process, we applied IScap on mouse meninges to activate the TG neuron axons. Less than 40% of WT mice show headache-like behaviors (using paws to wipe the top of head) in this headache model. Remarkably, more than 80% of KO mice display headache-like behaviors, indicating that loss of TRESK indeed increases the likelihood of getting headache. Furthermore, we used FG labeled TG neurons to specifically record the “headache-generating” neurons. Indeed, the “headache-generating” neurons from KO mice are more excitable than those from WT mice.
In addition to headache-like behaviors, TRESK KO mice showed more robust responses to painful heat, cold, touch and chemical stimuli on facial skin as well.
Interpretation
Having established that TRESK channels regulate headache and other facial pain, we then asked whether loss of TRESK affects DRG neurons and body pain behaviors in KO mice.
A previous study demonstrated increased DRG neuronal excitability and body pain behaviors in KO mice. Surprisingly, we found that none of the DRG neurons from our KO mice were hyper-excitable. We believe that unlike TG neurons, DRG neurons can upregulate other currents to fully compensate for the loss of TRESK-like current. This ensures that DRG neurons generate normal responses to painful stimuli without TRESK. Consequently, KO mice have normal behaviors to nociceptive heat, cold, touch, and chemical stimuli on hindpaws.
The discrepancy between our results and the previous work may arise from differences between genetic background and/or the housing conditions of the two strains of KO mice. If so, it suggests that nature and/or nurture influence whether defective TRESK channels affect body pain.
Visit eNeuro to read the original article and explore other content. Read other summaries of eNeuro and JNeurosci papers in the Neuronline collection SfN Journals: Research Article Summaries.
TRESK K+ Channel Activity Regulates Trigeminal Nociception and Headache. Zhaohua Guo, Chang-Shen Qiu, Xinghua Jiang, Jintao Zhang, Fengxian Li, Qin Liu, Ajay Dhaka, and Yu-Qing Cao. eNeuro July 2019, 6 (4) ENEURO.0236-19.2019; DOI: 10.1523/ENEURO.0236-19.2019








