How Misfiring of Striatal Cholingeric Interneurons Can Explain Early Cognitive Deficits in Huntington's Disease
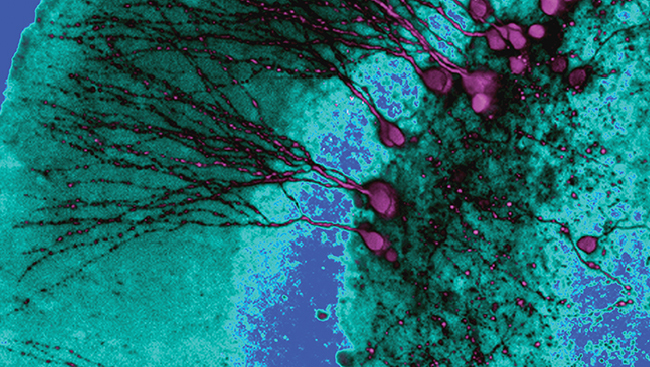
Material below summarizes the article Enhanced GABAergic Inputs Contribute to Functional Alterations of Cholinergic Interneurons in the R6/2 Mouse Model of Huntington’s Disease, published on February 24, 2015, in eNeuro and authored by Sandra M. Holley, Prasad R. Joshi, Anna Parievsky, Laurie Galvan, Jane Y. Chen, Yvette E. Fisher, My N. Huynh, Carlos Cepeda, and Michael S. Levine.
Huntington’s disease (HD) is an inheritable neurological disorder caused by a triplet repeat (CAG) expansion in the huntingtin (HTT) gene located in the short arm of chromosome four. Symptoms include uncontrollable dance-like movements (chorea), cognitive deficits, and psychiatric disturbances. Some of the early, or prodromic, symptoms include an inability to shift attention and other behavioral inflexibilities.
The principal pathological feature of HD is the loss of medium-sized spiny neurons (MSNs) in a region of the brain called the striatum. The striatum provides the major input into a larger area called the basal ganglia. Large cholinergic interneurons (LCIs) represent only 2-3 percent of the neurons found within the striatum; however, their impact on striatal function is enormous. Activation of thalamostriatal inputs leading to patterned LCI activity have been shown to be critical for attentional shifts, specifically the presynaptic suppression of cortical input mediated by muscarinic M2/M4 receptors and cessation of ongoing motor activity when relevant stimuli occur (Ding et al., 2010).
A reduction in cholinergic markers and acetylcholine (ACh) release has been demonstrated in patients and in mouse models of HD (Farrar et al., 2011). Reduced cholinergic markers can explain deficits in synaptic plasticity and behavioral inflexibility (Picconi et al., 2006) and possibly dysregulation of excitatory cortical input. Studies have shown that ACh, acting on muscarinic receptors in corticostriatal terminals, can regulate excitatory input onto MSNs (Pakhotin and Bracci, 2007). What leads to the cholinergic deficits is, however, unknown.
In eNeuro, Holley et al. unravel the possible causes of LCI dysfunction. In their study, the authors found evidence of abnormalities in morphology and membrane properties consistent with reduced somatic size of the LCIs in symptomatic HD mice. Importantly, this was found to correspond with reduced pacemaking ability of LCIs in these animals. In normal animals, most LCIs were found to fire regularly whereas in HD mice they displayed irregular firing patterns and more action potential bursts. While some of these changes can be explained by altered intrinsic ionic conductances, synaptic inputs also modulate firing of LCIs. In particular, GABAergic inhibition can disrupt firing of LCIs. Consistent with this observation, in their study, Holley et al. found that inhibitory inputs are significantly increased in LCIs in the brains of the symptomatic HD mice. In contrast, excitatory inputs remained unchanged.
To dissect the origin of increased inhibition, the authors used optogenetics and found that activation of somatostatin- but not parvalbumin-containing interneurons evoked larger amplitude inhibitory responses in LCIs from HD mice compared with the normal control animals. In contrast, glutamatergic spontaneous or evoked postsynaptic responses were not affected.
Based on these data, the authors concluded that morphological and electrophysiological alterations, in conjunction with the presence of mutant huntingtin within the LCIs, could explain impaired capacity for ACh synthesis and release in HD mice and possibly in human patients. Dysregulated glutamate release is a key feature in the early stages of HD (Cepeda et al., 2007) and it is likely that altered ACh transmission plays an important role. This has potential implications for the development of novel therapies for HD, towards finding ways to restore normal LCI firing patterns and cholinergic neurotransmission which could help treat behavioral and cognitive deficits associated with the disorder.
Visit eNeuro to read the original article and explore other content. Read other summaries of JNeurosci and eNeuro papers in the Neuronline collection SfN Journals: Research Article Summaries.
Enhanced GABAergic Inputs Contribute to Functional Alterations of Cholinergic Interneurons in the R6/2 Mouse Model of Huntington’s Disease. Sandra M. Holley, Prasad R. Joshi, Anna Parievsky, Laurie Galvan, Jane Y. Chen, Yvette E. Fisher, My N. Huynh, Carlos Cepeda, Michael S. Levine. eNeuro Jan 2015, 2 (1). DOI: 10.1523/ENEURO.0008-14.2015








