Material below summarizes the article Pathways for Emotions: Specializations in the Amygdalar, Mediodorsal Thalamic, and Posterior Orbitofrontal Network, published on August 26, 2015 in JNeurosci and authored by Clare Timbie and Helen Barbas.
In everyday life, emotional scenes captivate us, and we remember them more readily than ordinary scenes and events. Pathways that convey sensory signals reach neurons in the thalamus, which then convey the signals to the cerebral cortex, the brain’s outer rim. But the external world and its emotional import are intertwined in our experience. How does information about our inner world reach the cortex as we become aware of our emotions — happiness, sadness, fear?
The brain’s key center for emotions is the amygdala, which is hidden in the temporal lobe. Studies of pathways — the brain’s vast highway system — have shown that the prefrontal cortex receives information from the sensory areas of the cortex as well as from the amygdala. In particular, the orbitofrontal part of the frontal lobe is distinguished for its multimodal connections: it receives pathways from cortical areas that represent each of the sensory modalities and the amygdala. The orbitofrontal cortex thus has a panoramic view of the external sensory environment and the internal environment of emotions.
The amygdala communicates with the orbitofrontal cortex in more ways than one. A direct pathway goes straight from the amygdala to the orbitofrontal cortex. A second pathway was thought to communicate first with the thalamus (the mediodorsal nucleus), and from there with the orbitofrontal cortex. This indirect pathway resembles the passage of signals from our senses to the thalamus, and then to the sensory areas of the cortex. But until recently, we did not know if the same neurons in the thalamus that receive signals from the amygdala connect directly with the orbitofrontal cortex.
The reason for the uncertainly is that studying pathways from the internal world through the thalamus is not as clear-cut as it is for the sensory systems. The mediodorsal nucleus receives signals from structures in addition to the amygdala and communicates with areas in addition to the orbitofrontal cortex.
How can we be sure that the neurons that receive signals from the amygdala in the mediodorsal thalamus are the same neurons that communicate with the orbitofrontal cortex?
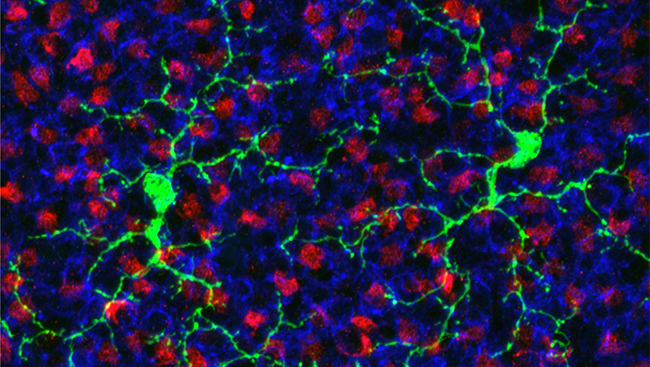
To solve this problem, it was necessary to inject small amounts of distinct tracers to map more than one pathway simultaneously. Neural tracers travel forward or backwards in neurons, or in both directions, depending on the tracer. Retrograde tracers travel backwards from the site of injection. When a minute amount (a few microliters) of a retrograde tracer is injected in one brain site, the dye is taken up by the axons of neurons in the vicinity of the injection site. The tracer (dye) travels backwards from the axons to the parent cell bodies, labeling the neurons and thus the pathway. In thin sections of the brain viewed under the microscope, we can see the labeled neurons whose axons reach the injection site. Anterograde tracers move forward in neurons — from the cell body all the way down the axon terminals. Under the microscope, we see the tiny terminals of the axons. The pathway mapped extends from the site of injection to the axon terminals.
We addressed the issue of whether a pathway from the amygdala through the mediodorsal thalamus to the orbitofrontal cortex was truly sequential by injecting a retrograde tracer in the orbitofrontal cortex; this tracer traveled backwards to label the cell bodies of neurons in the thalamic mediodorsal nucleus. In the same brain, an anterograde tracer injected into the amygdala traveled forward and labeled the axons and their terminals in the mediodorsal thalamus.
This double-labeling experiment revealed that the axon terminals from the amygdala overlapped in the thalamus with labeled neurons that project to the orbitofrontal cortex. Under a light microscope, it is possible to get a broad overview of these pathways. At higher levels of resolution in the confocal and electron microscope, it is possible to zero in at the sites of interaction of these pathways in the thalamus — the close contacts where information is passed from one neuron to the next.
These multiple approaches showed that the neurons that receive pathways from the amygdala in the thalamic mediodorsal nucleus project to orbitofrontal cortex, confirming the presence of two sequential pathways.
These experiments showed that the orbitofrontal cortex has at least two ways to receive information about emotions: through a direct pathway from the amygdala, and through an indirect pathway through the thalamus.
But are the direct and indirect pathways similar or different? Further study revealed that these pathways are distinct in several ways.
The neurons in the amygdala that project directly to the orbitofrontal cortex are small, while the ones that project to the thalamus are large, even though they are intermingled in the amygdala. The direct and indirect pathways also differ by their label for a specific transporter of the major excitatory neurotransmitter glutamate.
This analysis suggested that the pathway from the amygdala to the orbitofrontal cortex, which innervates most strongly the upper layers, has a modulatory influence. On the other hand, the pathway from the amygdala to the thalamus forms large synapses that suggest a strong influence on the thalamus. The pathway from the mediodorsal thalamus to the orbitofrontal cortex innervates the upper as well as the bottom layers of the cortex through parallel pathways that have distinct neurochemical features as well.
Taken together, these findings show that information about emotions from the amygdala has sure ways to reach all layers of the orbitofrontal cortex, a region with a premiere role in emotions and social interactions.
Visit JNeurosci to read the original article and explore other content. Read other summaries of JNeurosci and eNeuro papers in the Neuronline collection SfN Journals: Research Article Summaries.
Clare Timbie and Helen Barbas. (2015): Pathways for Emotions: Specializations in the Amygdalar, Mediodorsal Thalamic, and Posterior Orbitofrontal Network, The Journal of Neuroscience, August 2015, 35(34): 11976-11987; DOI: 10.1523/JNEUROSCI.2157-15.2015








