Transplantation of Healthy GABAergic Interneuron Progenitors in Mice with Epilepsy
Material below summarizes the article Restrained Dendritic Growth of Adult-born Granule Cells Innervated by Transplanted Fetal GABAergic Interneurons in Mice with Temporal Lobe Epilepsy, published on March 27, 2019, in eNeuro and authored by Jyoti Gupta, Mark Bromwich, Jake Radell, Muhammad N. Arshad, Selena Gonzalez, Bryan W. Luikart, Gloster B. Aaron, and Janice R. Naegele.
Highlights
- Transplanting healthy inhibitory cells into the dentate gyrus of the hippocampus in mice was shown to inhibit new granule cells generated after temporal lobe epilepsy (TLE).
- The inhibitory connections formed by the transplanted cells are linked to changes in the structure of new granule cells, including smaller dendritic arbors.
- Many of these adult-born cells become highly abnormal in TLE and contribute to the development of spontaneous seizures, so these structural changes may be important for reducing the excitability of new granule cells in TLE and offer hope to people who suffer spontaneous seizures and sometimes seek relief through brain surgery.
 |
|
|
Study Question
Throughout life, new granule cells, and their dendrites, generate from stem cells in the hippocampus, one of a few brain regions that maintain neurogenesis. Does transplanting GABAergic interneurons into hippocampus regulate the growth of new granule cells?
How This Research Advances What We Know
Unencumbered information flow and memory rely on granule cells in the hippocampus and their stereotypic dendritic arbors. Throughout life, new granule cells generate from stem cells in the dentate gyrus, one of a few brain regions that maintain neurogenesis.
Granule cell production is a delicate operation impacted by environment, exercise, or disease, including temporal lobe epilepsy (TLE). The impact of TLE on granule cell production can cause newborn granule cells to form highly abnormal dendritic arbors, migrate improperly, and sprout new connections. These structural changes enhance excitation, making seizures more likely. Additionally, seizures damage GABAergic interneurons and induce molecular and genetic alterations that contribute to hyperexcitability.
Effective treatment for people with drug-resistant forms of TLE includes resection of the damaged regions of the hippocampus, but bilateral removal causes severe memory impairment. That outcome led our group to explore alternatives including a promising approach being studied for many neurological disorders: neural stem cell transplantation.
In mice with TLE, seizures have been reduced following transplantation of human stem cell-derived GABAergic interneurons or fetal GABAergic progenitors. In this study, we investigated the synaptic connections formed by transplanted GABAergic interneurons onto adult-born granule cells and the effects this has on their dendritic arbors.
Experimental Design/Methodology
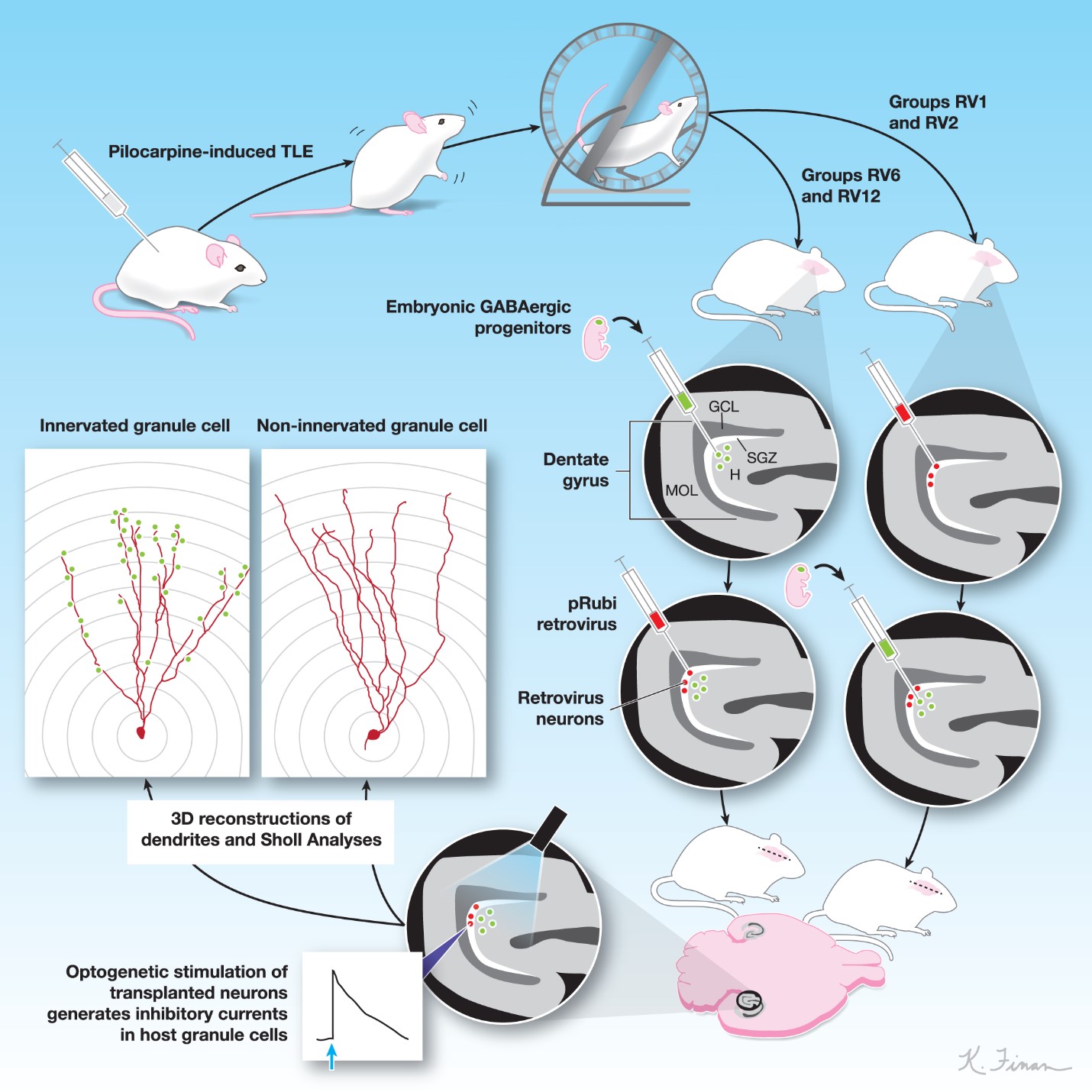
We harvested GABAergic progenitors on embryonic day 13.5 of gestation from transgenic mouse embryos expressing Channelrhodopsin (ChR2) under the control of the vesicular GABA promoter (VGAT-ChR2). The dissociated cells were transplanted into the dentate gyrus of each hippocampus in adult male mice with pilocarpine-induced TLE and into naïve mice.
At different times after transplantation, adult-born granule cells were fluorescently labeled by injecting a retrovirus into the subgranular zone. After maturation of the labeled cells, we studied their connectivity with the transplanted cells in hippocampal slices using optogenetics. We recorded from granule cells using patch-clamp electrophysiology, a technique in which fine micropipettes are attached to individual cells to study ion currents.
To test for transplant-induced synaptic inhibition, we delivered brief pulses of light to depolarize the transplants, while recording post-synaptic currents in the adult-born granule cells. We later compared the dendritic arbors of retrovirally labeled granule cells that exhibited transplant-derived inhibitory post-synaptic currents to those that did not show transplant-derived inhibitory post-synaptic currents.
We quantified the size of the dendritic arbors and lengths of branches in over one hundred adult-born granule cells in adult mice using three-dimensional reconstructions, created with IMARIS software and Sholl analyses, a method for quantifying dendritic arbor sizes and branch lengths.
Results
While recording the electrophysiological activity of adult-born granule cells labeled with the retrovirus, we found that exciting the transplanted cells optogenetically rapidly induced inhibitory post-synaptic currents in many adult-born granule cells. This result indicates that in some cases, functional inhibitory synapses are formed by the transplanted cells directly onto adult-born granule cells.
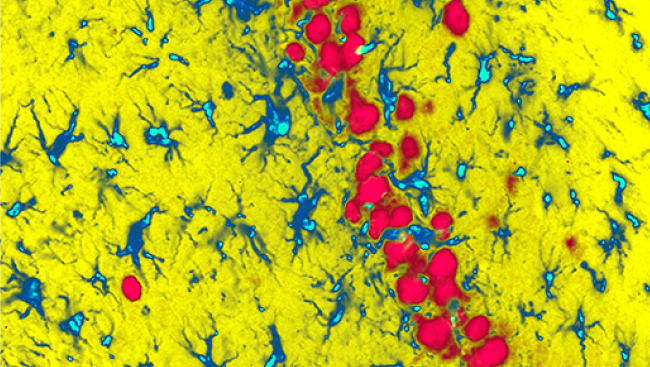
High-resolution confocal images of adult-born granule cells also showed high numbers of transplant-derived axonal swellings on the cell bodies and dendrites of adult-born granule cells, suggesting the transplants formed synaptic contacts.
Quantitative analyses comparing the dendritic arbors of adult-born granule cells with or without transplant-derived innervation showed that connectivity with the transplants was linked to significantly smaller dendritic arbors in adult-born granule cells and overall reductions in their total dendritic lengths. These effects of the transplants on dendritic arbor size in adult-born granule cells were remarkably persistent, as the effects were found even with delays of weeks or months between transplantation of GABAergic progenitors and birth of new granule cells.
Interpretation
Hippocampal granule cells are important for regulating information flowing into the dentate gyrus of the hippocampus, and their stereotyped conical dendritic arbors are related to their function. The growth-inhibiting effect of the transplanted progenitors on adult-born granule cell dendrites is observed in both epileptic and naïve mice, and only in granule cells that show high levels of input from the transplanted interneurons. This observation indicates restricted dendritic growth occurs through local synaptic mechanisms, rather than by a more general mechanism such as seizure suppression.
Moreover, the transplanted cells are capable of altering the growth of dendritic arbors in granule cells born weeks or even months after transplantation, suggesting long-term effects of the transplanted cells on successive populations of adult-born neurons added to the dentate gyrus. Future work may help to clarify the molecular signals regulating the growth of adult-born granule cell dendrites and how reducing dendritic arbor size alters their functional connectivity and intrinsic properties.
Visit eNeuro to read the original article and explore other content. Read other summaries of eNeuro and JNeurosci papers in the Neuronline collection SfN Journals: Research Article Summaries.
Restrained Dendritic Growth of Adult-Born Granule Cells Innervated by Transplanted Fetal GABAergic Interneurons in Mice with Temporal Lobe Epilepsy. Jyoti Gupta, Mark Bromwich, Jake Radell, Muhammad N. Arshad, Selena Gonzalez, Bryan W. Luikart, Gloster B. Aaron, and Janice R. Naegele. eNeuro March 2019, 6 (2) 0110–18.2019; DOI: 10.1523/ENEURO.0110-18.2019








